- Home
- Weddings
- Portraits
- Journal
- Contact
- R shankar quantum solution
- Celtic kings rage of war ghul
- Listen to the secret of kells soundtrack
- Element 3d v2-2 crashes after effects cc
- Gtr2 wsgt
- Winchester 94ae 44 mag value
- Chocolatier 3 game online
- Big sean and jhene aiko
- Crack for moho pro 12
- Free word stack game
- Magic screen magnification software price
- Depriester chart bubble p
- Jriver - hqplayer integration
- Autodesk maya 2015 windows 10

Read this K-value off the chart (approximately 21.3).
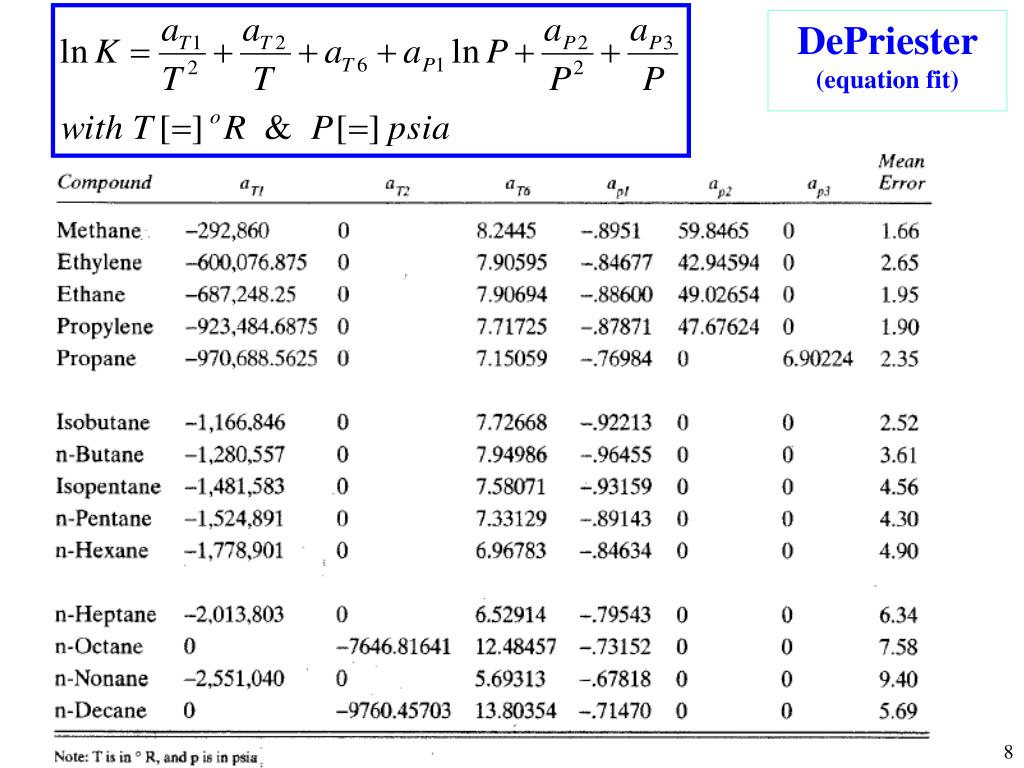
Depriester chart bubble p manual#
Wankat (Instructors Solution Manual) SOLUTION MANUAL for SEPARATION PROCESS ENGINEERING.
